AIDE: Week Six
Early 20th Century plastics timeline
Notable (abbreviated) steps along the way
Courtesy of the Plastics Historical Society
- 1284
-
The Horners' Company was first recorded in 1284 in the City of London.
The Horners Company was awarded a Royal Charter in 1638.
Horn is deposited in laminated layers around a bony core separated by connective tissue. It grows throughout the life of the animal, whereas antler is shed each year.
One method was to soak the horn in barrels of water for a specific time depending on the season. When removed from the barrels the horn was heated before splitting with a sharp knife, prising open with tongs and putting between hot metal plates in a press and applying pressure.
Placing the warmed horn into moulds before applying pressure enabled the production of horn boxes, beakers and other items.
By 1725 London was a major centre for moulding of horn.
 Ricard Gibon Anno Domini 1612 in the Horner’s Company collection.
Ricard Gibon Anno Domini 1612 in the Horner’s Company collection. - 1656
-
Gutta Percha (GP)
1656
John Tredescant introduced Gutta Percha from the Far Eastern Palaquium tree bark. This produced a hard material mouldable by heat to soften.
 Gutta Percha golf balls - Circa 1894 (Valued at $2,200)
Gutta Percha golf balls - Circa 1894 (Valued at $2,200)Gutta Percha is thermoplastic, softening at elevated temperatures and returning to its solid form as it cools. This made it easy to mould into many decorative and functional objects, either by pressing the heated material into cold moulds, or by extrusion.
-
Properties
1846 - R. and J. Dick Ltd., founded in Glasgow, are still making belting from GP.
Electronics
Gutta-percha latex is biologically inert, resilient, and is a good electrical insulator with a high dielectric strength.By 1845, telegraph wires insulated with gutta-percha were being manufactured in the United Kingdom. It served as the insulating material for some of the earliest undersea telegraph cables, including the first transatlantic telegraph cable. Gutta-percha was particularly suitable for this purpose, as it was not attacked by marine plants or animals, a problem which had disabled previous undersea cables.
Polyethylene's superior insulative property has displaced it.
Dentistry
The same bioinertness that made it suitable for marine cables also means it does not readily react within the human body. It is consequently used in a variety of surgical devices and for dental applications during root canal therapy.
It is the predominant material used to 'obturate', or fill the empty space inside the root of a tooth after it has undergone endodontic therapy. Its physical and chemical properties, including but not limited to its inertness and biocompatibility, melting point, ductility, and malleability, afford it an important role in the field of endodontics. -
Other uses
1851 - The Great Exhibition featured over 100 applications of GP.
In the mid-19th century, gutta-percha was also used to make furniture, notably by the Gutta-Percha Company (established in 1847). Several of these highly ornate, revival-style pieces were shown at the 1851 Great Exhibition in Hyde Park, London. Molded furniture forms, emulating carved wood, were attacked by proponents of the design reform movement, who advocated truth to materials.
The material was quickly adopted for numerous other applications. The "guttie" golf ball (which had a solid gutta-percha core) revolutionized the game. It was gradually replaced with superior (generally synthetic) materials, though a similar and cheaper natural material called balatá is often used in gutta-percha's place.
- 1820
-
Rubber and derivatives
1820-1943
Thomas Hancock investigated intensively the moulding of masticated rubber and its many uses.
1843
Hancock refined and patented vulcanisation, establishing the UK rubber industry with products including boats, springs, hoses and bands. Vulcanite (hard rubber or ebonite) was an important innovation.1851
Nelson Goodyear (USA) patented ebonite primary thermosetting material for demanding applications such as battery boxes, pumps, telephones, dental plates and fountain pens.
1861
Francis Shaw introduced the first screw extruder for rubber production.1900
Synthesised methyl rubber poly(dimethylbutadiene)1915
Synthetic methyl rubber production began in Leverkusen1930
Nitrile rubbers (BUNA) produced in 1939 in the USA1931
Du Pont (USA) produces polychloroprene rubber (Neoprene)1937
Urethane rubbers introduced by R. M. Thomas ((USA)1939-40
Goodrich develops polybutadiene and styrene-butadiene rubbers which made major contributions to World War II1943
Silicone polymers were introduced by Dow Corning Corporation - 1850's
- 1870's
- 1892
- 1900's
-
Cellophane
1905
J. Edwin Brandenberger (USA) invented cellophane used in thin films for fabrics and packaging providing major applications for many years.
-
Synthetic Plastics
1910-1920
1910 Formica electrical insulation laminate by H. Faber and D O'Conor (USA) using p/f resin impregnated paper plies.
1912 I. Ostromislensky (Russ.) patented the polymerisation of vinyl chloride to give unstable PVC polymer (unstable when not modified).
 Blue Amberol Records are made of blue celluoid around a plaster of paris core, and play for 4 minutes at 160 rpm. Their introduction led to a resurgence of sales of cylinder records.
Blue Amberol Records are made of blue celluoid around a plaster of paris core, and play for 4 minutes at 160 rpm. Their introduction led to a resurgence of sales of cylinder records.1913 Klatte produced polyvinyl acetate.
1918 Hans Johns patented urea-formaldehyde resins.
- 1920
-
Synthetic Plastics
1920-1930
1922 Herman Staudinger (Ger.) proposed long chain molecular structures for polymers and synthesised rubber.
1924-28 Edmund Rossiter developed water-white transparent mouldings from thiourea-formaldehyde resins (marketed as Beetle resins from 1928).
1926 The first truly successful commercial injection moulding machine was produced in 1926 by Eckert and Ziegler (German Patent 495362).
1927 Otto Rohm (Ger.) developed poly (methyl methacrylate) clear plastic.
 1927: Gestetner duplicating machine featuring Bakelite casings designed by Raymond Loewy
1927: Gestetner duplicating machine featuring Bakelite casings designed by Raymond Loewy
-
Synthetic Plastics
1930-1940
1927-37 Wallace Carothers headed major Du Pont team into 'designed' macromolecules, e.g. Neoprene rubber introduced in 1931 followed in 1934 with Nylon fibre (Nylon 66; Hill).
1931 Heidrich (Germany) produced the first screw extruder specifically for thermoplastic processing.
1938 Nylon 6 fibre (Schlack, I. G. Farben) commercially produced as Perlon.
1932 ICI introduces PMMA - Perspex
1933-40 Henkel patented melamine production for melamine formaldehyde resins. Developed by Ciba, American Cyanamid as basis for moulding powders (Melaware) and M-F surfaced laminates (Formica).

1933 W. Semon produced stable PVC polymer (Goodrich).
1933 Dow discovers polyvinylidene chloride (Saran).
1933 First injection moulded polystyrene articles produced following earlier work by BASF and Dow Chemicals (produced in scale in 1937).
1933 ICI discovers low density polythene, full commercial production in 1939.
1933 Unsaturated polyester resins, developed and introduced commercially into the USA in 1941. (Cold-cured styrenated polyesters introduced in 1946 for fibre reinforced plastics.)
 Charles and Ray Eames, Eiffel Tower base chair, 1948-1950.
Charles and Ray Eames, Eiffel Tower base chair, 1948-1950.1937 Hans Kellerer (Austria) introduced a fully automatic injection moulding machine capable of continuous operation.
.jpg) Fada Baby radio - Catalin is transparent, near colorless, rather than opaque.
Fada Baby radio - Catalin is transparent, near colorless, rather than opaque.1938 Du Pont discovers PTFE.
1939 Bayer introduces polyurethane systems.
- 1939
-
Space Age Materials
1939-1950
1939 Foundation epoxy resins. 1945 Ciba produced epoxy resins.
1940 Du Pont introduced Polyacrylonitrile (PAN) polymer, an early engineering product.
1940 PET - poly(ethylene-terephthalate) synthesised by J. R. Whitfield and J. T. Dickson at Calico Printers Association. ICI Terylene fibres and Melinex films followed (also Dacron and Mylar from Du Pont). In 1977 oriented blow moulded drinks containers were launched based on PET polymers and other types, e.g. PBT - poly(butyl-terephthalate).
1947 Formica melamine faced decorative p/f laminates were introduced to the UK.
 1957 Melmac Idealware by Russel Wright. A brand name of dinnerware molded from melamine resin, made by American Cyanamid, most popular in the 1940s through the 1960s.
1957 Melmac Idealware by Russel Wright. A brand name of dinnerware molded from melamine resin, made by American Cyanamid, most popular in the 1940s through the 1960s. -
Space Age Materials
1950-1960
1953 Karl Ziegler with E. Holzkamp produced high molecular weight, low pressure poly(ethylene) using organo-metallic catalysts; a major step forward.
1953 Bayer and General Electric independently produced polycarbonate pioneering engineering polymer.
1954 Isotactic polypropylene discovered by Guilo Natta, Montecatini. ICI produced 'Propathene' used in high volumes for demanding domestic and engineering applications.
1955 Du Pont patented EVA (ethylene-vinyl acetate copolymer) with the Elvax range launched in 1960.
1959 Du Pont launched Delrin - polyformaldehyde polymer (Delrin 100TL gear mouldings shown below). This proved to be a valuable light engineering polymer.
- 1960
-
Space Age Materials
1960-1970
1961-62 Du Pont introduced polyimide films - Kapton - and varnishes and later foams for composite structures. The resins are capable of high insulation, fire resistant and mechanical properties.
1964 Polyphenylene oxide (PPO) polymers were patented by General Electric (USA) and introduced as the Noryl range in 1966. These were widely used as high performance engineering thermoplastics.
1965 Union Carbide, 3M and ICI produced polysulphone thermoplastic engineering polymer.
1965 PEEK - Poly(ether.ether ketone) resistant chemical and engineering polymer produced by ICI.
Courtesy www.sme.org
1966 vinyl ester polymers (bisphenol A/acrylic variants) produced by Dow Chemical Co. as the Derakane series.
 Designed by Eero Aarnio in 1967, the "Pastilli Chair" is also known as the Gyro or Pastil chair.
Designed by Eero Aarnio in 1967, the "Pastilli Chair" is also known as the Gyro or Pastil chair.Carbon Fibre Dragonfly Drone - assisted by vinyl esters. Festo by way of Composites Today
-
Further Reading
1970's onward
Resource The Plastiquarian
The First Century of Plastics - Celluloid and its Sequel Morris Kaufman 1961; (Fundamental landmark publication.)
Fibre Composite Hybrid Materials ed. N. L. Hancox; Applied Science Publishers, 1981. (High performance polymers, adhesives and reinforcements etailed for design and moulding of composites for demanding applications, e.g. prosthetics.)
- On the shoulders of these developments and greater understanding of control, structure, manufacture, design and test techniques has engendered a proliferation in space and electronic age applications.
Comparisons illustrated
World Ressource Institute (WRI) illustrates the activity sectors from which of greenhouse gases (GHGs) originate, comparing this world flow chart with that of the USA.
'All data is for 2000 and given in CO2 equivalents with the GWP 100a weighting factors for methane, nitrous oxides, HFCs and PFCs from the IPCC 1996 report. The total quantity is an estimate of 41755 MtCO2 equivalent.'
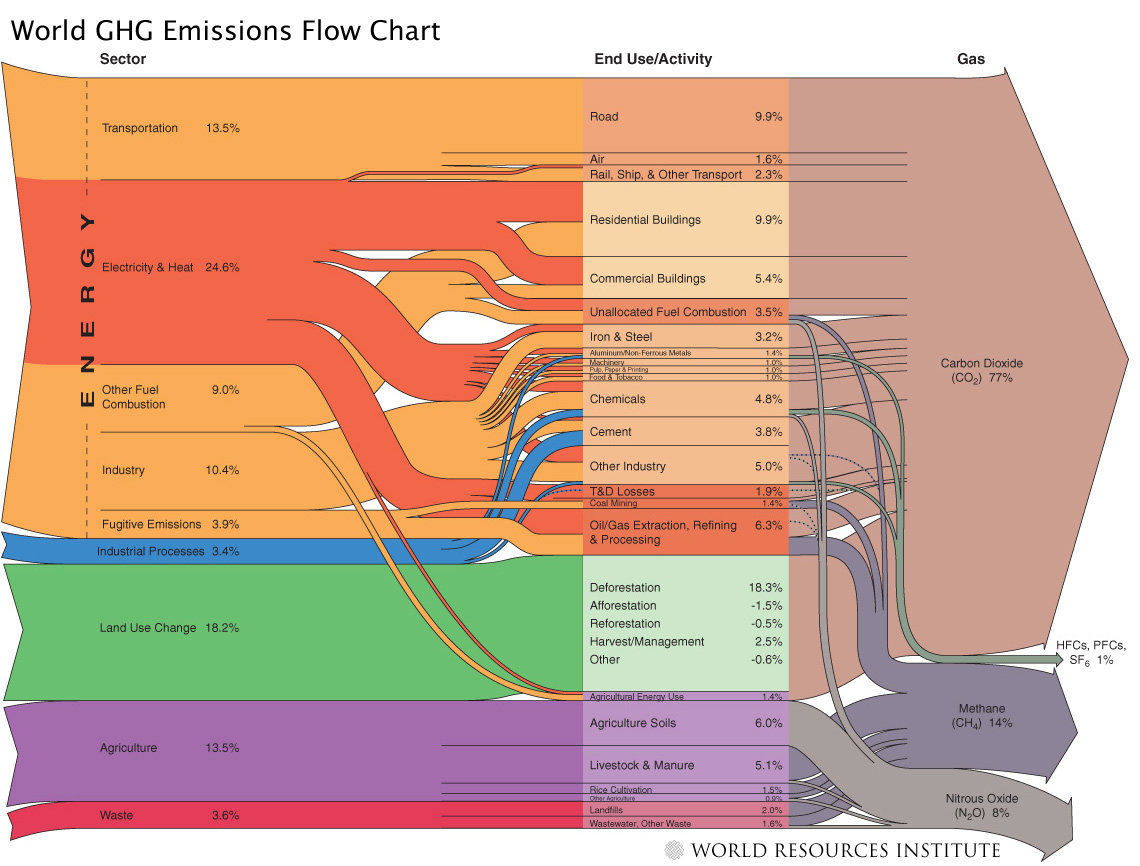
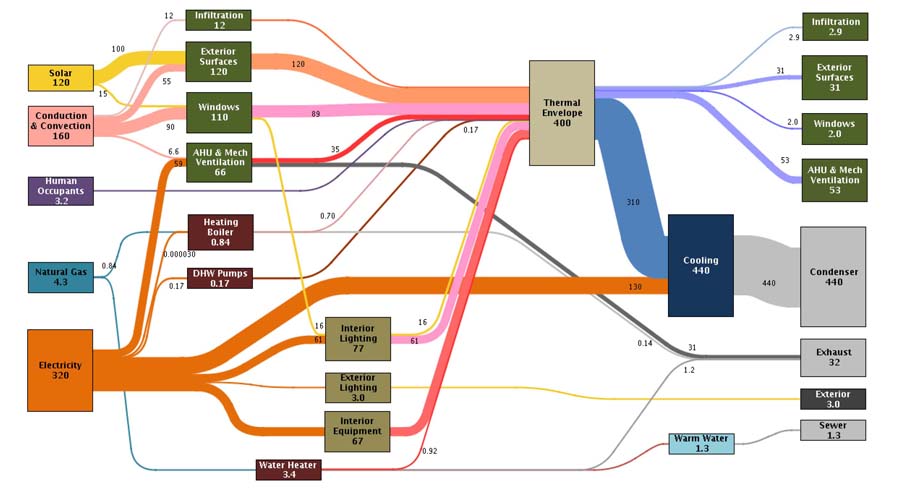
Sankey Diagrams
This article on ‘A Pilot for Measuring Energy Retrofits’ describes how researchers from the EEB Hub used an old navy building in Philadalphia to “determine detailed system performance”.
The result of that “inverse modelling” (i.e. measuring) approach are presented in Sankey diagrams and are used “to identify discrepancies in the predicted versus actual energy balance”.
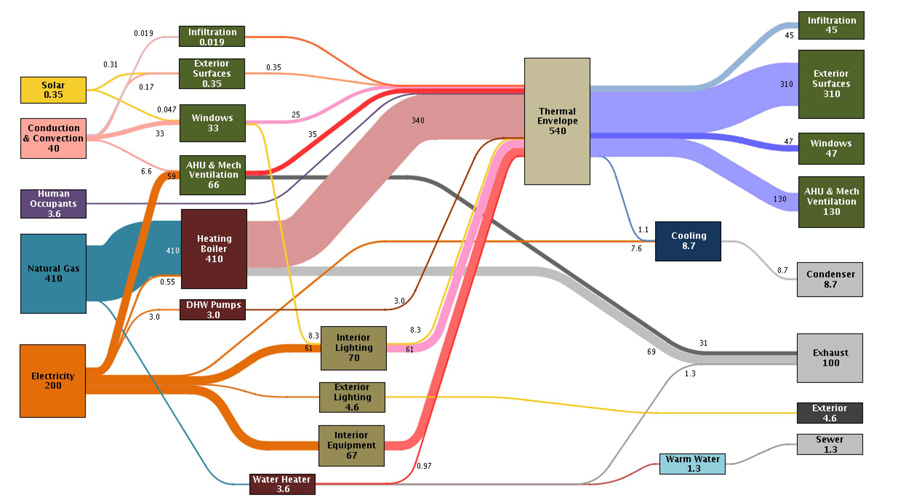

4see
This short film introduces the 4see framework whose purpose is to explore future scenarios using data from the past. Foresight
Student example
A simple example illustrating Sankey Diagram application.
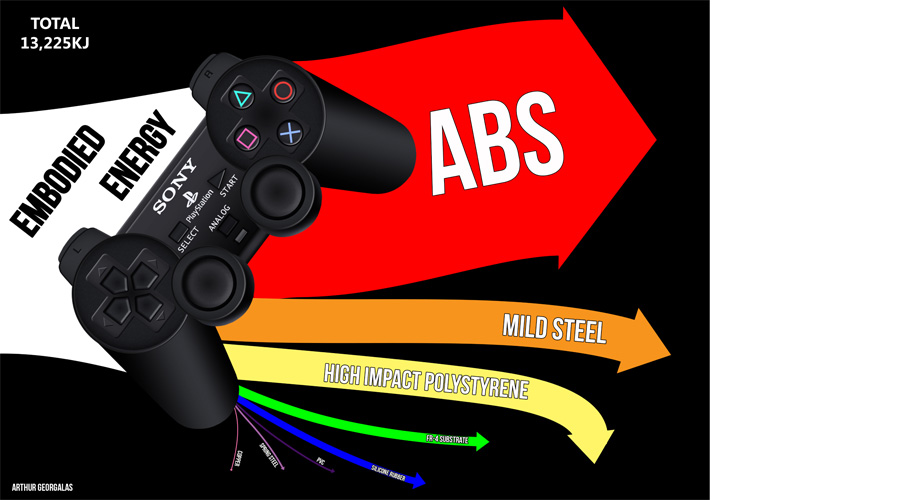
Student example
The modified product embodied energy.

Pathways
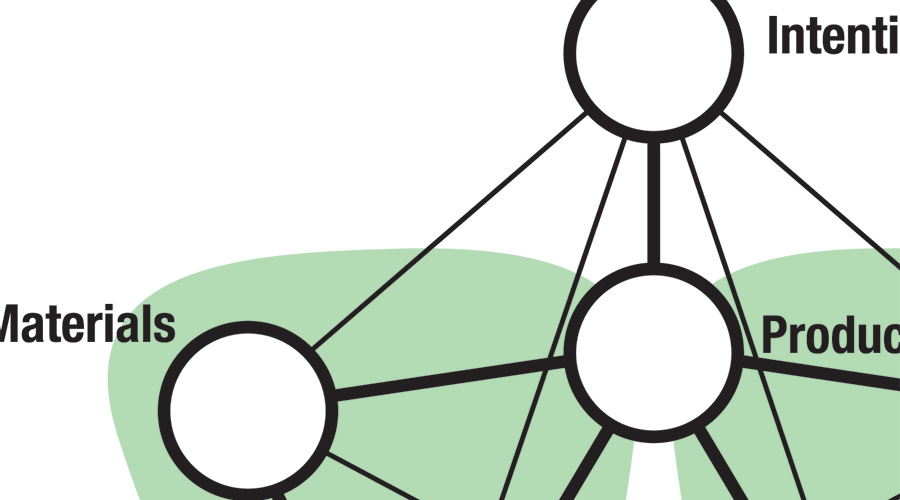
'Makers' consistently congregate and support one another's survival. The flow of energy, material, parts and artefacts can be mapped within these manufacturing precincts and beyond in the distribution phase.
A geographical map will act as a foundation for the following LCA mapping.
Download Plastics Presentation pdf
Assignment 2: Sustainability Analysis
Analyse the autopsy product discussing sustainability principles relating to your product.
Present the key components in a clear illustrative discussion, including:
The Materials life-cycle map
Map the source and eventual destinations of each autopsy material as pathways and intersections.
Discuss briefly the origins of the autopsy product within the context of the mapping result.
Postulate on the next generation within the context of the mapping result.
Manufacturing nodes
Discuss your group versions highlighting the comparative materials and manufacturing idiosyncracies.
Describe the assembly processes as annotated nodes on the map.
Include discussion about re-assembly and re-use by visualising the end-of life pathways on your map.
Table your findings.
Discuss the value of the product
Think of yourself as a consumer versus you as a designer.
Think about where the product was manufactured, the culture and environment the product was designed for and the implied product life cycle pathways of the end user.
Poll ten people on their perspectives on the product. Map their pathways of usage upon your map.
Table your findings
-
Discuss the end-of-life issues relating to the key autopsy components.
Present your findings at class in Week 9. Submission
Compile your reflections and data as a Case Study including iterative modifications of assignment One plus the Assignment Two components.
Present your Case Study mapping submission in Week 10.